Virus-Like Particles (VLPs) are biomolecular nanoparticles formed by controlled self-assembly of viral structural proteins. Lacking viral DNA for replication, VLPs are capable of triggering an immune response without the risk of infection and have been proven to be safe yet highly immunogenic. VLPs are of growing interest in the pharmaceutical and biotechnology industries following the success of blockbuster VLP-based vaccines against hepatitis B and human papillomavirus. These bioparticles are increasingly targeted not only for safe and efficacious immune stimulants but also therapeutic gene delivery agents, taking advantage of their DNA encapsulation and transport capabilities.
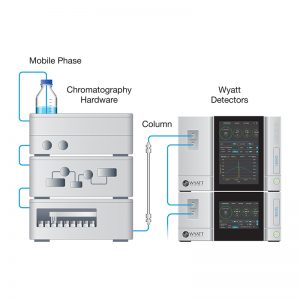
With an increasing number of VLPs entering biotherapeutic development pipelines, accelerated development presents several key analytical challenges. Common to all phases of development is the basic biophysical characterization of mass, size, composition and conformation. These and additional, phase-specific properties are required in early stage stability assessment of vaccine candidates; process development; and multiparametric formulation development. In the final product, quality assurance requires identifying and quantifying degradant products such as free capsomeres, partially formed capsids and aggregates.