Analysis of purity and demonstration of lot to lot comparability is essential throughout the drug development process. Among the various biophysical and biochemical assessments of purity to which biological therapeutics are subjected, one of the most fundamental is based on the molecular weight of the species present in aqueous solution.
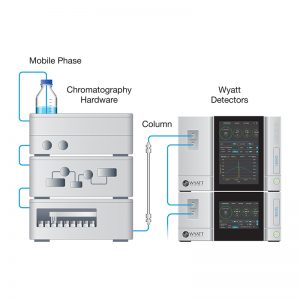
A common approach to evaluate the relative levels of molecular weight forms is to perform analytical size exclusion chromatography (SEC) in combination with multi-angle light scattering (MALS) and absorbance (UV) and/or refractive index (RI) concentration detectors. For monoclonal antibodies, a dominant peak (>95% total peak area and molar mass consistent with an intact antibody) along with a small number of minor high and/or low molecular weight species is frequently observed. While SEC-MALS data are robust and precise, the method is not without potential concerns. Sample dilution, weak non-specific interactions with the column, shearing, or exclusion of high molecular weight species are frequently cited effects that may impact the complete analytical characterization of purity based on size fractionated material.