Interactions between proteins and nucleic acids often result in binding stoichiometries greater than 1:1 and may exhibit cooperativity, allosteric hindrance, or other complex phenomena. Quantifying the affinity and stoichiometry of these biomolecular assemblies is key to understanding the mechanism of interaction and to developing therapeutics that target and modulate them.
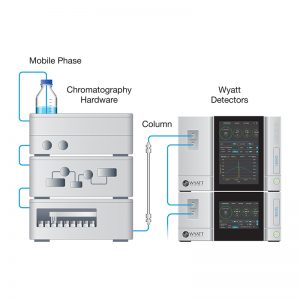
Here, we present the characterization of protein-DNA complexes using two complementary multi-angle light scattering (MALS) techniques: 1) in combination with size exclusion chromatography (SEC-MALS) and protein conjugate analysis to measure the overall complex molar mass and fraction of DNA and 2) composition-gradient MALS (CG-MALS) to quantify the stoichiometry and affinity at each binding site in the complex formation.