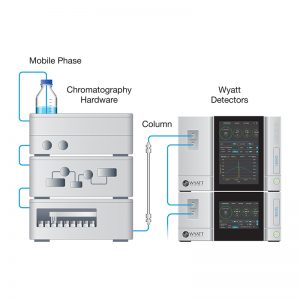
Quantifying binding affinity between species with multiple binding sites can present a significant challenge to many biophysical characterization techniques. Composition-gradient multi-angle light scattering (CG-MALS) provides direct measurement of affinity and absolute stoichiometry for a wide variety of interactions, including multivalent interactions, without the need for surface immobilization or tagging.
A model fusion protein, Y, was engineered with two binding sites for its ligand, X. The interaction was quantified using two CG-MALS experiments. The first experiment quantified the interaction between X and Y at a highaffinity binding site and suggested the presence of a second low-affinity binding site. A simulation based on these results led to the design of a follow-up experiment to quantify the affinity at the weaker binding site and confirm that no additional higher order species were formed.