The adeno-associated virus (AAV) is an attractive delivery vehicle in gene therapy1,2 attributed to its mild immune response and ability to deliver its genetic payload into a wide range of host cells. The first FDA-approved AAVbased gene therapies are Luxturna® by Spark Therapeutics and Zolgensma® by Novartis for treating a rare genetic eye disease and spinal muscular dystrophy, respectively. With these approvals and many other AAVmediated in vivo gene therapy drug candidates in clinical trials, it is essential that robust and reliable characterization tools are implemented in order to understand the quality attributes of this class of therapeutic products, ensuring their safety and efficacy.
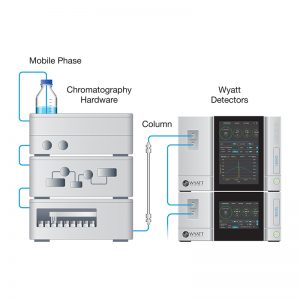
A size-exclusion chromatography (SEC) method using triple detection–UV, differential refractive index (dRI), and multi-angle light scattering (MALS)–has been developed to measure the following three important AAV quality attributes (QAs): 1) Total number of viral capsid particles; 2) Relative capsid content (e.g., ratio of empty and full capsids); and 3) Percentage of monomer or aggregates.